Turnkey cleanroom modules
Valicare supports you in bringing your cell and gene therapies to production on time.
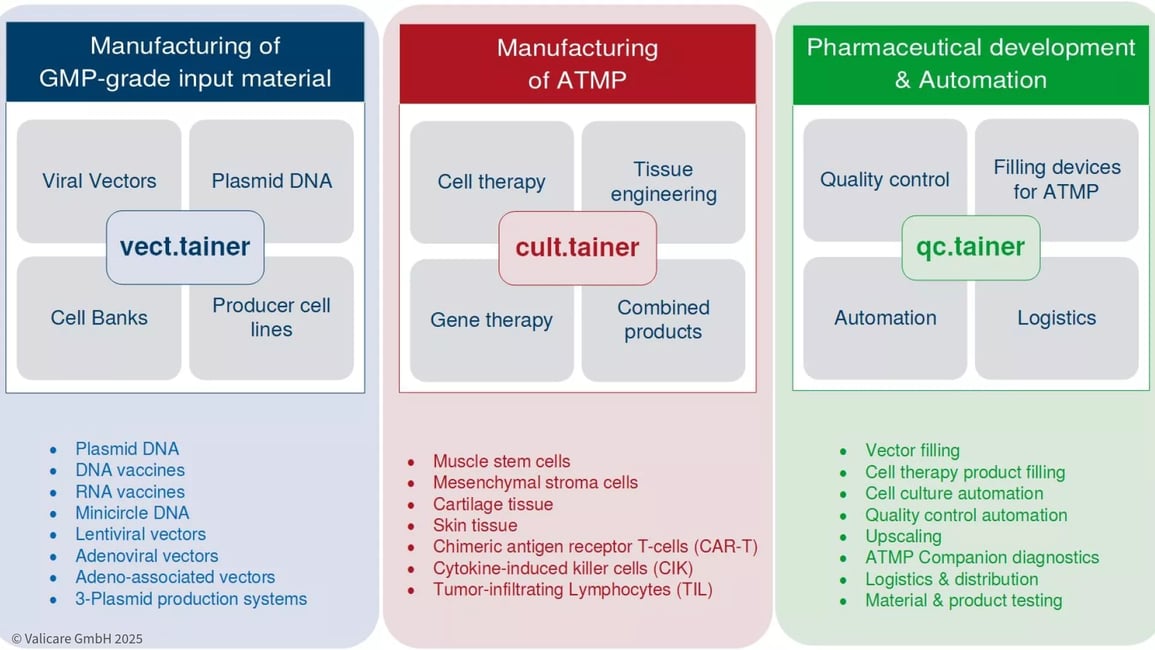
Advanced therapy medicinal products (ATMPs) is an overall term for gene therapies, cell therapies and biotechnologically processed tissue products. The number of clinical trials and approvals for these is increasing rapidly on a global scale. In the future, their enormous potential will enable therapies for a wide range of diseases which were previously uncurable.